Don't Be Afraid of the Glow in the Dark!
Fall is in full swing and Halloween is approaching. It’s the time of year for glowing ghosts, ghouls, and… science experiments!
Things that appear to glow are luminescent. Luminescent materials are literally “cool” because they give off light without needing or producing heat. Luminescence can be broken down into the following main categories: fluorescence, phosphorescence, and chemiluminescence.
Fluorescent materials will absorb energy, then quickly re-emit the energy. As a result, they only appear to “fluoresce” when they are in the presence of some form of radiation such as ultraviolet light.
The PASCO Spectrometer allows you and your students to experiment with fluorescence. Fluorescein, as the name implies, is a chemical that will exhibit fluorescence. In this demonstration, a small sample of fluorescein is diluted in water, then added to a cuvette. When held under a blacklight (ultraviolet radiation source) the sample will glow. In the Spectrometry App under Fluorescence, we can set an excitation wavelength to 405 nm.
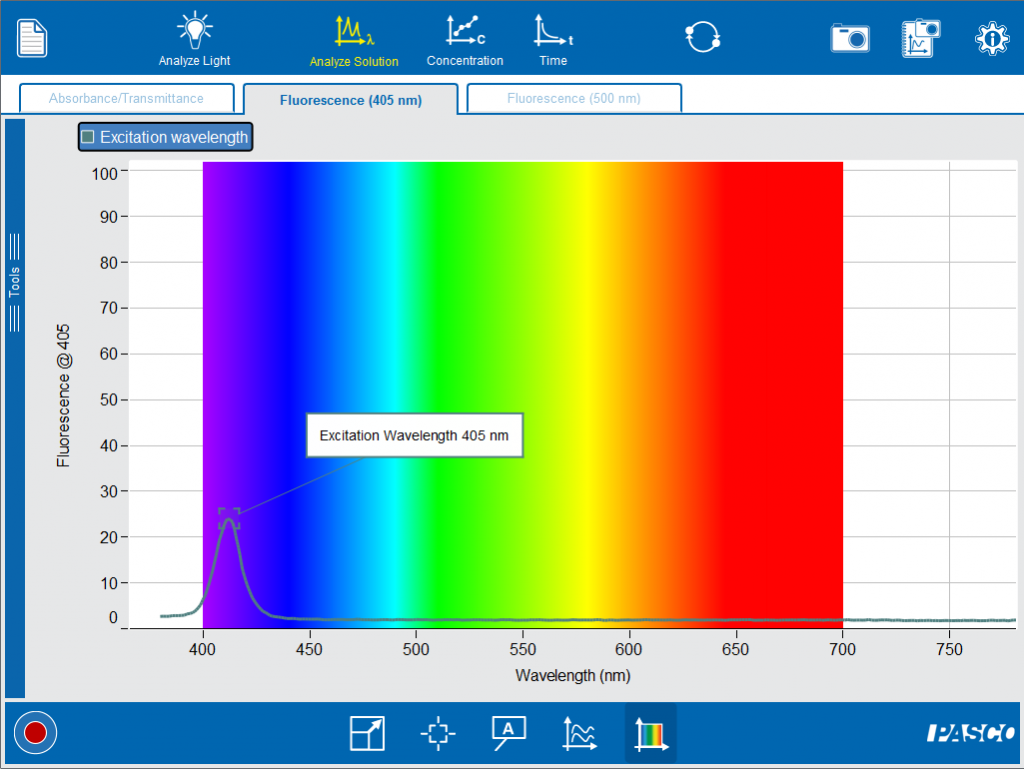
When the cuvette with fluorescein is added to the Spectrometer, you can observe the “glow” indicating fluorescence.

Now we can observe the spectrum of the emitted light when fluorescein is excited with 405 nm light.
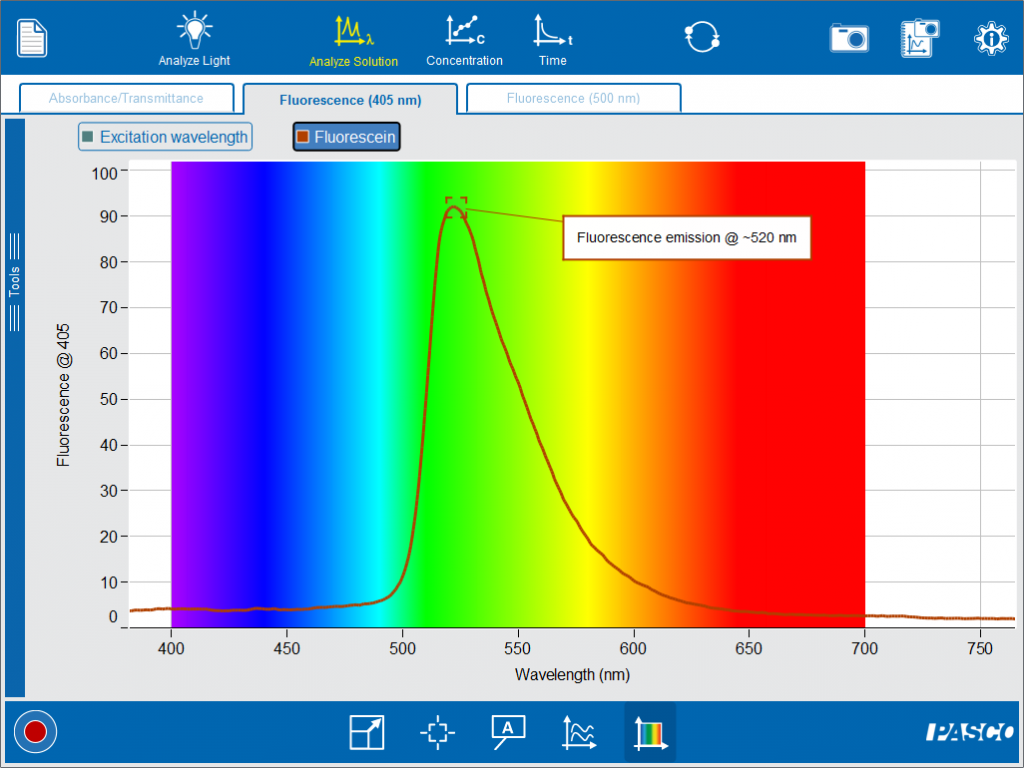
By overlaying the spectra, we can compare the wavelength of the light that went into the sample and the light that was fluoresced by the sample.
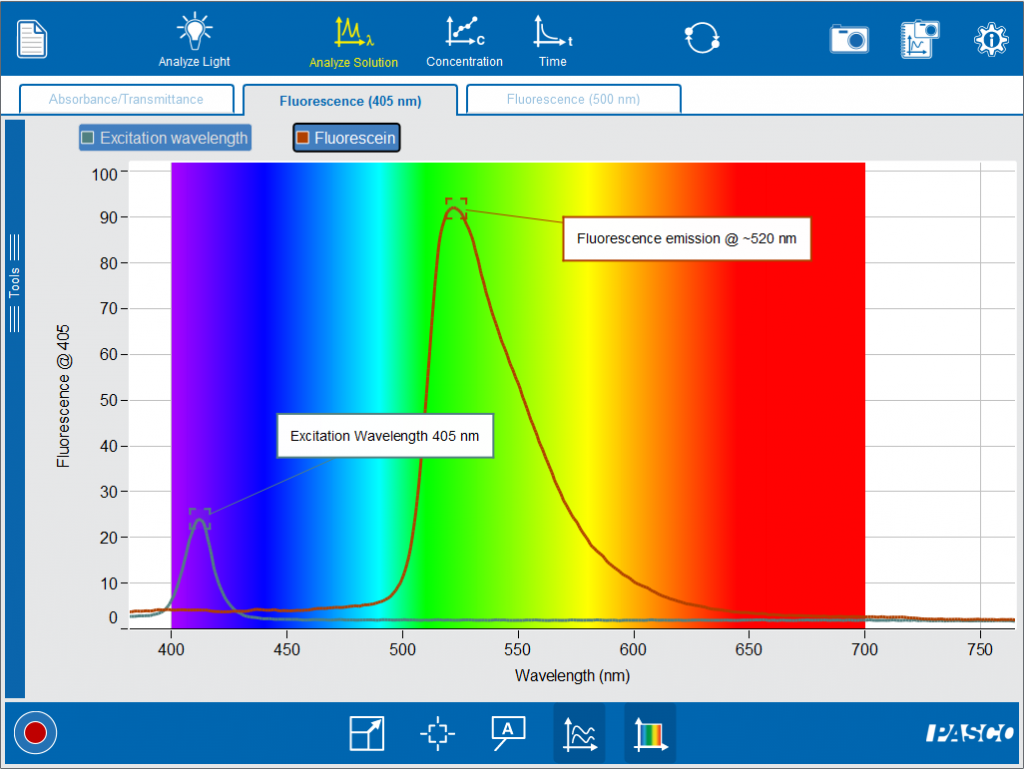
Phosphorescent materials glow in the dark. Similar to fluorescence, they get excited by white or ultraviolet lights. But these materials slowly re-emit the energy in the form of light, even when the lights are turned off. Glow-in-the-dark toys are a great example of phosphorescence.
Finally, chemiluminescence occurs when a chemical reaction produces light without producing heat. Glow sticks are a perfect Halloween example of this. When the chemicals are mixed, a ghostly glow is given off.
So, the next time you see a glowing jack-o-lantern or an eerie zombie, don’t just think scary… think science.

